How basic behavior of LENR can guide a search for an explanation
How basic behavior of LENR can guide a search for an explanation – Revised by Edmund Storms LENRGY LLC Santa Fe, NM, 87501 (4/2/16)
download pdf
ABSTRACT
The LENR effect was identified 27 years ago by Profs. Fleischmann and Pons as production of extra energy by a normal chemical structure, in this case PdD. Over a thousand published papers now support the discovery and the energy is shown to result from fusion of hydrogen isotopes without the need to apply energy and without energetic radiation being produced.
By conventional standards, the claims are impossible. Nevertheless, a new phenomenon has been discovered requiring acceptance and understanding. The major behaviors and their present understanding are described in this paper and are used to suggest how an effective explanation might be constructed. Once again, science has been forced to either reject the obvious or accept the impossible.
In this case, the normal skepticism needs to be ignored in order to determine if this promised energy source is real and can provide the ideal energy so critically needed.
INTRODUCTION
Low Energy Nuclear Reaction (LENR) or Cold Fusion was introduced to the world 27 years ago by Fleischmann and Pons(1), Univ. Utah, with expectation of great benefit to mankind. Instead, their claim for a new kind of fusion was quickly rejected (2), an attitude that continues even today. Over the years, several thousand papers addressed the subject with a large fraction supporting the claim(3). Mastery of about 1000 papers is now required to understand the effect.
A description of all the known behaviors and all proposed explanations would require much more than a single review paper. Here, only the tip of the large iceberg will be examined along with some original results not published elsewhere. The selection of behaviors is designed to focus attention on only the essential conditions required to cause the LENR effect.
Limits will be set using observed behavior in order to evaluate proposed explanations. The new kind of nuclear interaction needed to explain LENR is expected to fall within these limits. In other words, boundaries need to be identified to keep the imagination from running wild. The LENR effect is assumed consistent with all rules normally applied to conventional chemical and nuclear behavior. Nevertheless, a novel mechanism is clearly operating and needs to be acknowledged.
Many conditions needing consideration are not quanitative or lend themselves to mathematical analysis. While frustrating to conventional scientists, these unique behaviors must be made part of a successful explanation. Quantitative behaviors can be used to expand understanding once the basic process is understood.
The present paper has two parts, with the first describing the important observations on which an explanation must be based. The second part uses a few assumptions combined with these chosen behaviors to provide an explanation about how LENR can be initiated and the resulting mechanism.
The LENR mechanism is clearly much different from that causing the conventional hot fusion process. Ironically, this conflict is used to reject the claims for LENR rather than guiding a search for the cause of the difference. This difference must be clearly understood before the novel features of LENR can be explored. Consequently, the hot fusion process is discussed first.
Unlike hot fusion, LENR takes place in and requires a chemical structure to operate. The role of this structure must be understood before physics is applied to understanding subsequent nuclear process. Clearly, a unique and rare condition must form in the structure in which a nuclear process can function. The nature of this condition is discussed following the discussion of hot fusion.
The nature of the hot fusion mechanism
Because LENR involves fusion of hydrogen, the conventional fusion process, called hot fusion, needs to be understood in relation to LENR. For the last 75 years, the hot fusion method has been applied in various ways, including in the ITER(4) facility now being constructed in France using magnetic confinement and in the National Ignition Facility(5) in Livermore, CA with lasers being used to create the required energetic plasma. These methods use high energy to overcome the Coulomb barrier by brute force.
This large applied energy changes the fusion rate in plasma as shown by the log-log plot in Fig. 1. The energy applied to LENR is no more than 1 eV.
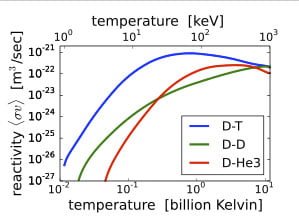
FIGURE 1. Effect of energy on the fusion rate in plasma for different combinations of hydrogen isotopes as result of the hot fusion process. (Wikipedia)
Hot fusion can also be initiated by bombarding a material by energetic deuterons. In this case, the fusion rate is slightly greater at low applied energy compared to when the same energy is applied to plasma, as can be seen in Fig. 2. Even so, the overall fusion rate
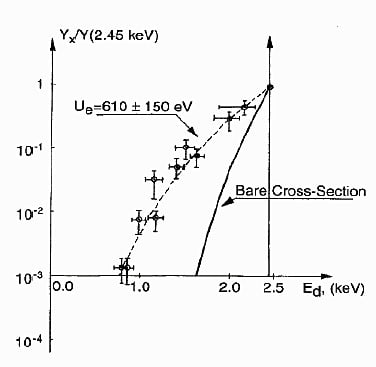
FIGURE 2. Comparison between the fusion rate in plasma (Bare Cross-Section) and when fusion occurs in a solid material as the result of applying energy to the bombarding D+ ions, as shown by the X-axis. A value of unity occurs when the rate in plasma is equal to the rate using a target material.(6)
decreases as applied energy is reduced. In other words, the environment in a material can slightly increase the fusion rate but it does not significantly offset the reduction in the rate as applied energy is lowered. While the electrons clearly help lower the barrier to achieve hot fusion, this effect alone would seem too small to explain the LENR process, although it might make a small contribution. In any case, the measured shielding effect applies only to the hot fusion mechanism.
Perhaps more effective shielding during LENR might be expected if the shielding electrons were contained in the unique nuclear-environment rather than having a random and lower concentration in the general where hot fusion interaction takes place. An evaluation of just how the electrons function during LENR compared to hot fusion requires LENR not be viewed as extension of hot fusion.
Once the nuclei of deuterium have fused by hot fusion, the assembly breaks into fragments, which dissipate the excess mass-energy as kinetic energy. Easily detected energetic neutrons, tritium, protons, and He3 are produced in equal amounts. This process is understood and is consistent with conventional expectations. A similar result occurs when muons are used to bring the nuclei close enough to cause fusion. In other words, no matter whether energy is used to overcome the Coulomb barrier by brute force or the separation is reduced by using the heavy muon(7-10), the same energy dissipation process results.
No other method for energy dissipation as result of a fusion reaction was known to occur in nature until “cold fusion” was discovered. Clearly, the mechanisms causing hot fusion and cold fusion are significantly different because LENR does not lead to fragmentation of the nuclear products.
Cold fusion is novel because it does not require significant applied energy to overcome the Coulomb barrier and it does not result in fragmentation of the fusion product as occurs during hot fusion. This difference has caused much skepticism about the reality of LENR. After all, experience and teaching deny any possibility of spontaneous fusion taking place in an ordinary chemical structure without the need to apply significant energy.
This apparent contradiction is resolved by proposing the cold fusion process takes place in a unique structure, called the nuclear-active-environment (NAE) where a novel mechanism can operate. Questions about how this structure forms, where in the chemical structure this formation takes place, the nature of the unique conditions at the NAE, and the nuclear mechanism operating therein are explored later in this paper.
Role of chemical structure
Because the LENR process takes place within a chemical structure, it must play by the rules such a structure imposes. This conclusion is critical to understanding the LENR process. These rules include the Laws of Thermodynamics and the Phase Rule. Local energy cannot spontaneously increase without violating the Second Law of Thermodynamics and the local concentration of ambient energy is limited by how much energy the chemical bonds can tolerate before melting or decomposition results. Simply stated, energy cannot go up hill and its density cannot exceed the strength of the container.
If a novel mechanism is proposed to concentrate energy in order to cause nuclear fusion, why it is not found to affect chemical reactions? After all, if such a process were possible, it would be expected to operate in normal chemicals and cause chemical effects before the local energy had increased enough to cause a nuclear reaction.
For example, the mechanism of energy transfer to electrons proposed by Widom and Larsen(11, 12) would be expected to make many normal chemical compounds unstable. Furthermore, how such a proposed violation of the Second Law of Thermodynamics can function in PdD needs to be justified. Similar conflicts with the laws of thermodynamics and normal chemical behavior create a similar weakness in many explanations now being proposed.
Normally, nuclear reactions of any kind are not affected by the chemical environment because the energy states are too different and local energy density cannot be increased according to the Second Law of Thermodynamics. Amazingly, the normal level of local ambient energy is sufficient to initiate the LENR process at high rate on rare occasions. Explaining how this “magic” takes place is the first of two basic challenges. The second challenge involves how the resulting energy is dissipated as heat.
Once fusion occurs, the structure must convert the excess mass-energy to heat without causing local melting. After all, local destruction of the active site would stop further heat production and severely limit the amount of energy produced by LENR, which is not experienced.
Although local melting is occasionally seen, it is not sufficient to create a limit to the amount of power or its stability over time. Thus, both the presence of a little local melting and the absence of extensive melting have to be explained.
Several different chemical structures have been found to support LENR, with PdD given the most attention. Consequently, PdD is the focus of further discussion.
Palladium deuteride has attracted interest for about the last 100 years(13) during which time it has been studied extensively. Although the palladium can acquire hydrogen up to about PdD0.98±0.02, nothing about the overall behavior would suggest an ability to host a fusion reaction.
The structure is face-centered-cubic (fcc) and exists in two slightly different forms having the same crystal structure based on the Pd sublattice. The alpha phase occurs between pure Pd and about PdD0.05, and the beta phase forms near PdD0.6 when 1 atm of D2 pressure is applied at 20° C. A two-phase region exists between these two compositions. The beta phase continues to acquire D atoms at random sites in the fcc sublattice as pressure is increased, finally reaching the upper limit of the fcc phase.
Fig 3 shows the structure when all lattice sites are fully filled by deuterium. Another phase is expected to form and grow in amount as the overall D/Pd ratio increases beyond the upper limit to the fcc phase, similar to the behavior of other metallic hydrides.(14, 15) In other words, any composition in excess of PdD0.98 would be expected to be a two-phase mixture of the fcc and another phase having a different structure and increased stoichiometry.
In the absence of the rare double occupancy(16, 17) of normal lattice sites, the deuterium nuclei are too far apart to fuse. Achieving close approach without violating the rules of chemistry and without producing fragmentation typical of hot fusion remains a serious challenge discussed in a later section.
Identifying where the NAE is located and what form it takes in the material has created a problem for many proposed explanations. Many explanations assume the fusion process takes place in a modification of the fcc structure when the D/Pd ratio is large. Formation of such a structure would be apparent because its formation would cause changes in various properties.
A search for the expected change can be made by examining several known properties, such as resistivity and lattice parameter as a function of D/Pd. The lattice parameter can be seen to have a linear(18-21) relationship to composition with no indication of a two-phase region forming within the limits of the beta phase. Both the pressure and resistivity(22) also show no sign of a change in crystal structure(23) over the composition range of interest. In every way, all properties are consistent with a normal fcc structure being present within the composition range in which LENR is found to occur.
FIGURE 3. Crystal structure of the face-centered-cubic PdD when all deuterium sites (small purple) are filled. (Wikipedia)
On the other hand, Fukai(24) reported formation of a phase change when high pressure is applied at high temperature to PdH. This structure is proposed to also form under normal conditions during electrodeposition.(25) Superabundant vacancies are proposed to form in the metal sublattice. A similar structure change is proposed to be caused by deformation induced vacancies.(26) This behavior might also occur when repeated loading and deloading of PdD causes the structure to expand, producing what Storms(27) calls excess volume. Nevertheless, this condition does not explain LENR because the presence of excess volume over about 2% is found to inhibit LENR(28) rather than aid the reaction as would be expected if formation of metal atom vacancies were required to support LENR.
Even though the proposed vacancies are not associated with the LENR process, a unique condition is expected to form in the PdD in order for LENR to take place. This conclusion is consistent with common experience. When a piece of Pd is found to be nuclear active, most of the entire batch is also found to be nuclear active.
In addition, once the sample is made nuclear active, the LENR process using that piece becomes reproducible and robust. Obviously, treatment of the entire batch of Pd creates stable conditions in which the LENR process can be initiated and supported for extended times.
Unfortunately, these conditions are hard to produce because their unique characteristic is unknown and rarely formed. Even when certain important initial conditions are present, an additional special treatment is required before the nuclear process can be produced by PdD. These observations are important because they show a treatment is possible to make large amounts of palladium nuclear active. A suggested combination of conditions is described later in this paper.
Initially, the LENR reaction was thought to take place anywhere in the PdD structure. Later studies reveal both helium(29, 30) and tritium(31) form only very near the surface and not within the bulk material or on the surface where nanoparticles might be present when electrolysis is used. Transmutation products are also detected mainly in the surface region.
Based on the known behavior of helium in PdH(32, 33), the nuclear reactions apparently take place within a region perhaps no more than 10 μm wide, extending from the surface. We now need to discover the nature of the unique condition forming within this narrow band. The condition does not appear to involve a phase change, creation of vacancies in the hydride structure, creation of nanoparticles on the surface, nor does it require a high concentration of deuterium.
Formation of NAE would appear to require conditions formed by a unique process, which apparently only forms near the surface.
IMPORTANT OBSERVED BEHAVIOR
Formation of the NAE
In order for fusion to take place, the reacting nuclei must obviously be in the same place at the same time. This condition is not normally present. Normally, the D atoms are located too distant to fuse.
For atoms to assemble in a chemical structure, Gibbs energy must be released while the material achieves a different stable state. Generally, the atoms in a chemical structure are close to their equilibrium condition and do not contain excess energy or have the ability to form another crystal structure unless the conditions are significantly changed. Simply increasing the D/Pd ratio does not create sufficient energy to change the structure in order to initiate the LENR process.
Furthermore, for the process to be as rare and as difficult to initiate as is observed, the conditions for releasing this energy must be equally rare and difficult to create. To make the problem even more challenging, once the NAE is formed, LENR must operate at a significant rate without further change in conditions. These conditions immediately place a limit on any proposed condition in which LENR can take place.
Most samples of PdD do not host the LENR process regardless of the deuterium content presumably because the unique NAE is not initially present in the material. This conclusion suggests the NAE is not related to any of the features normally found in a chemical structure, such as vacancies, dislocations, and occupancy of unusual lattice sites. After all, if the NAE were related to these common features, the effect would be initiated more easily and more often.
Multiple occupancy of the normal deuterium-atom vacancy must also be rejected based on this conclusion because, if such occupancy were possible, it would be present in all material under normal conditions and cause LENR with greater frequency.
Nevertheless, a rare condition must form as result of some kind of treatment in order to account for occasional success. Failure to initiate LENR simply means this treatment was not successful in producing the required NAE. Once produced, the NAE appears to be stable and relatively constant in amount as indicated by production of relatively constant power.
Experience reveals another important behavior. When part of a batch of palladium can be made nuclear active, the remainder of the batch is found to be active. This activation treatment does not simply involve reaction with D but also requires extended electrolysis and/or repeated deloading and loading with D.
This behavior is important because it revels a condition can be created throughout an entire batch of Pd as result of a common treatment that can eventually host the LENR process. In other words, the physical treatment before reacting with deuterium affects later initiation of LENR.
Once the nuclei are assembled in the NAE, a unique process must reduce the Coulomb barrier perhaps by a tunneling mechanism without using energy beyond that which is normally available at room temperature. Immediately, we are confronted by a problem. Normal chemical structures are known not to support nuclear reactions without significant energy being applied to bombarding ions. After all, the Coulomb barrier keeps nuclei separated and allows chemical structures to form in the first place by interaction between the electrons.
The energy required to force the nuclei close enough to fuse is well in excess of the energy holding the atoms in the structure and in excess of the electron energy. This well-known and accepted behavior suggests a need to form a novel
arrangement between the nuclei in the NAE designed to avoid this limitation.
In summary, two separate processes have to be considered. The first is creation of the NAE. The second is formation of a structure of H and/or D within the NAE having the ability to fuse. This nuclear process is separate from the structure of the NAE, but needs to be consistent with it.
A description of the fusion process is a job for physics while identification of the NAE is a job for chemistry. Thus, we are forced to acknowledge an uncomfortable marriage between two normally independent branches of science, with chemistry being applied first to identify the NAE.
Nature of the NAE
Two different kinds of NAE have been suggested. Many researchers place the LENR process in the normal crystal structure where vacancies or dislocations might be present. Different variations of the crystal lattice are proposed, including formation of nanoparticles and active sites on the surface of the structure.
In contrast, Storms(34) places the NAE in cracks having a critically small gap, which are separate from and chemically independent of the crystal structure. Such an environment can have properties much different from a crystal structure, including a high negative charge.
Resolving this fundamental difference in proposed location of the NAE is critical to understanding the LENR process because the chosen location sets the logic on the correct path to discovering the mechanism. A choice of the wrong path will result in arriving at the wrong understanding.
In order to contrast these two proposed conditions, the well documented suggestion by Hagelstein et al.(35) is explored. The Hagelstein idea is based on formation of a new phase in the normal fcc structure, such as suggested by Fukai and Okuma(36). This phase is proposed to form on occasion after deuterium content has exceeded D/Pd=0.85, thereby causing formation of palladium atom vacancies.
Deuterium atoms fill the vacant sites and form a structure in which fusion is proposed to occur. The resulting mass-energy is dissipated by phonons.
Evidence for this proposed phase change could be obtained by searching for a discontinuity in the various properties. As noted above, such a search reveals no evidence for a phase change within the composition range of the beta phase.
In addition, X-ray and neutron diffraction studies of the face-centered-cubic structure reveal no phase change in this composition range. Using a similar argument, all the other explanations of LENR involving changes in gross structure can be rejected.
The NAE is apparently a feature outside of the thermodynamic behavior and its presence does not affect the measured physical properties. While arguments based on the absence of behavior are usually ignored, in this case the failure of the physical properties to respond to the change required to form a NAE is an important characteristic of the LENR process.
The author, in several previous papers (37-39), proposes the NAE resides in nanocracks resulting from stress relief. These gaps exist outside of the chemical properties and are not influenced by the limitations imposed by the chemical structure.
As long as a gap having a critically small width is created, deuterons are proposed to enter the gap and form a structure that can be described in many different ways. This structure then experiences fusion by a novel mechanism.
The required gap width is rarely created because most cracks would quickly become too wide to host the required hydrogen structure. Success in creating the NAE involves creating modest stress and applying it to a structure containing many weak regions having similar ability to form small cracks.
This condition might be created by accident as result of various intended and accidental treatments applied during a study, thus accounting for occasional success without apparent reason. Although large cracks are often seen when LENR occurs, the cracks having the ability to act as the NAE are too small to be easily detected and can be overlooked.
In fact, unless these structures are sought using high magnification, they would be impossible to detect. Experience shows the critical initial condition can also be created in a batch of material by a yet to be identified pretreatment. This realization encourages the search for such a treatment from which production of large amounts of nuclear active material can be expected to result.
Deciding which explanation should be explored is important because they each propose entirely different treatments to cause the LENR process. The wrong choice of explanation can lead a researcher down the wrong path with much wasted effort.
Power production
The LENR effect was first identified by its ability to produce energy in amounts greater than would be possible by any chemical reaction. This energy has been produced when Pd is used as the cathode in an electrolytic cell using an electrolyte consisting of D2O+LiOD.
When a Pd cathode is initially subjected to this treatment, the deuterium concentration in the Pd increases while energy is absorbed by the reaction, as shown in Fig. 4. Energy is absorbed because the energy used to decompose the D2O into D2 and O2 is greater than is recovered when the resulting D2 reacts with Pd, thereby causing an overall endothermic reaction.
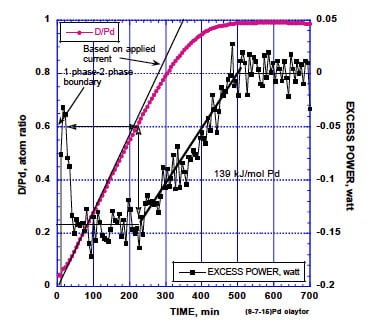
FIGURE 4. The D/Pd ratio and resulting power when Pd is reacted with D2O using the electrolytic method. All D made available by the applied current initially reacts with the Pd. The amount reacted is reduced only gradually as the upper limit is reached. No excess energy is produced even after the average D/Pd ratio becomes very large. The total amount of energy/mole Pd absorbed by the process is noted. (Storms, www.LENRexplained.com)
The enthalpy of formation for deuterium can be calculated using the data in Fig. 4. For this purpose, the total amount of D reacted every six minutes is divided by the amount of energy absorbed during this time, from which the amount of energy used to decomposed the D2O is subtracted.
As can be seen in Fig. 5, the electrolytic method applied to a solid piece of Pd gives values for the partial enthalpy of formation similar to the values obtained when D2 is reacted directly with Pd nanopowder. Both reactions show that chemical energy is released when Pd reacts with D2 and the amount decreases as the D/Pd ratio increases.

FIGURE 5. Enthalpy of formation calculated using the data shown in Fig. 4 based on the amount of D reacted every 6 minutes, the amount of power measured during this time, and the amount of energy used to decompose the D2O from which the D results. The reaction of D2 with Pd is exothermic. The Sakamoto et al. (40) line is obtained using their reported linear equation, which is then extrapolated from D/Pd= 0.85 to 0.98, and their reported D2 pressure. The pressure of D2 is also obtained from the review by Santandrea and Behrens(41). (Storms, www.LENRexplained.com)
The equilibrium deuterium activity, as pressure, is also plotted to show the large range in values being applied to the material. This quantity can be described as pressure only when the gas phase is present and is in equilibrium with the solid. The deviation from ideal behavior, called fugacity, is not taken into account.
No excess energy was produced even though a very high D/Pd ratio was reached. Additional treatment was later required to start the LENR process.
No additional phase forms in this composition range, such as proposed by Fukai, as indicated by the smooth unbroken variation of ΔH and pressure.
Also, the smooth unbroken change in resistivity observed by McKubre et al.(22) while LENR took place is consistent with this conclusion.
The effect of temperature on power production for various D/Pd ratios is compared in Fig. 6. Samples having D/Pd = 0.80 and 0.48 produce the same amount of power at the same temperature. Removal of all deuterium stops power production. Clearly, power is not as sensitive to the deuterium content as previous studies suggest(42). Nevertheless, some D is required for LENR to function.
The Arrhenius plot (Fig. 7), using the data in Fig. 6 (D/Pd=0.8), shows the activation energy for the LENR process to be nearly equal to the value for diffusion of D in PdD. In other words, the rate of the fusion process is sensitive to the rate at which D can get to the site where fusion takes place and it is not sensitive to the concentration of D in the surrounding lattice.
The fusion process can be proposed to rapidly convert deuterium in the NAE to fusion products, after which new D has to move relatively slowly from the surrounding lattice in order to supply additional fuel to the active sites.
The rate of energy production is determined by the rate at which D can get to the NAE.
By analogy, this is similar to the speed of a car being determined by how fast gas is delivered to the engine and not related to the amount of gas in the tank or the reaction rate within each cylinder. The resulting equation allows the resulting power to be predicted when temperature is increased.
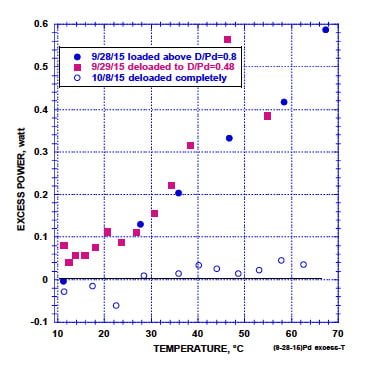
FIGURE 6. Effect of temperature on power production when three different amounts of deuterium are present in the sample. (Storms, www.LENRexplained.com)

FIGURE 7. Comparison between the rate of diffusion of D in PdD and production of LENR power as a function of 1/T. The similar slopes created by the data suggest both processes are affected by the same mechanism, i.e. diffusion of D though PdD. (43)
Probability of forming the NAE
Figure 8 compares power produced by 157 studies reported before 2007. Notice that most studies produce power at relative low levels. On a few occasions, a large amount of power is observed with the number of reports rapidly decreasing as the reported power increases. The number of reports, shown in Fig. 8 can be compared to
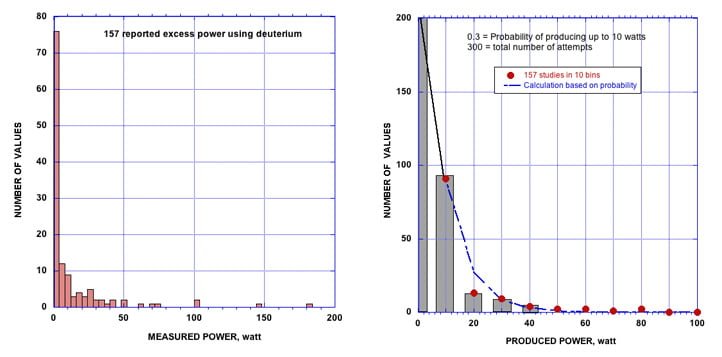
FIGURE 8. Histogram of power production vs the number of reported values. A probability function, shown as the dashed line, is used to fit the data to bins at 10 watt intervals.
predicted behavior based on an assumed probability of causing increased power once power production is possible. In other words, the probability of forming additional NAE once the conditions allow some NAE to form can be estimated and compared to the behavior to see if the assumption fits.
If 300 attempts are made to initiate LENR and the probability of producing 10 watts is 0.3, the probability of producing 20 watts would be 0.3×0.3, and the probability of producing 30 watts would be 0.3×0.3×0.3 etc.
The number of predicted successful observations at each power level is shown by the dashed line. The relatively good fit to the observed behavior suggests the power is caused by an increasing number of active sites whose production is caused by a random process, with more power resulting as the number of NAE sites is increased by a process having low probability. The probability of producing any power at all would be expected to be much less than producing additional power once conditions allow some NAE to form.
Continue reading How basic behavior of LENR can guide a search for an explanation
…
How basic behavior of LENR can guide a search for an explanation pdf TOC
INTRODUCTION
The nature of the hot fusion mechanism
Role of chemical structure
IMPORTANT OBSERVED BEHAVIOR
Formation of the NAE
Nature of the NAE
Power Production
Probability of forming the NAE
Helium Production
Tritium Production
Transmutation Production
Radiation Production
SUMMARY OF BEHAVIOR
CREATING A THEORY
ASSUMPTIONS
LENR Initiation as a Chemical Reaction
Nuclear Process Applied to LENR
Role of h4 formation
Consequence of LENR using a mixture of d and p
Transmutation
How does the fusion process work?
Reduction of Coulomb barrier
Dissipation of excess mass energy
Photons as the energy dissipation method
Electrons as the energy dissipation method
Phonons as the energy dissipation method
Storage of energy after fusion
Effect of different variables
Effect of laser irradiation
TESTABLE PREDICTIONS
Creation of the NAE
DISCUSSION
SUMMARY
